Abstract
Mantle cell lymphoma (MCL) is an aggressive form of NHL where frequent relapse following standard therapies remains a serious concern, even for promising new treatments such as combinations of a BTK inhibitor with the selective Bcl2 inhibitor venetoclax. Previous studies have also shown that MCL cells develop resistance through tumor microenvironment interactions that increase levels of Mcl1, BclxL, and/or Bfl1. Given the ability of CDK9 inhibition to deplete Mcl1 and Bfl1 (Boiko et al 2021), we explored the potential of clinical-stage inhibitor AZD4573 to induce apoptosis in MCL cell lines and PDX models as a monotherapy or in combination with acalabrutinib. Currently, AZD4573 is being evaluated as a monotherapy in a first-in-human study for relapsed or refractory hematological malignancies (NCT03263637) as well as in combination with the BTK inhibitor, acalabrutinib, in patients with non-Hodgkin lymphoma (NHL) (NCT04630756).
CDK9 is a serine/threonine kinase that mediates transcription elongation via phosphorylation of serine 2 of the RNA polymerase II carboxyl-terminal domain (pSer2-RNAP2). As previously shown, potent and selective inhibition of CDK9 by AZD4573 results in reduction of pSer2-RNAP2 levels leading to preferential depletion of labile proteins, including the Bcl2 family anti-apoptotic proteins Mcl1 and Bfl1 (as well as other known oncoproteins like Myc). This in turn drives rapid induction of apoptosis in a broad range of preclinical cancer models, particularly those derived from hematologic malignancies (Cidado et al 2020).
Here, we used 7 MCL cell lines and 1 PDX organoid to assess the rapid apoptogenic potential of AZD4573 in vitro. Cleaved caspase-3 (CC3), a hallmark of apoptosis, was measured immediately following acute treatment (6h) using Caspase-Glo 3/7. Four models were sensitive to CDK9 inhibition (EC 50 < 100nM; max. CC3 > 50%) while 1 cell line exhibited intermediate sensitivity (EC 50 < 100nM; max. CC3 < 50%) and 3 others were resistant (EC 50 > 100nM; max. CC3 < 50%). Regardless of sensitivity, AZD4573 caused a dose- and time-dependent reduction of pSer2-RNAP2, Mcl1, and Myc, consistent with our prior reports.
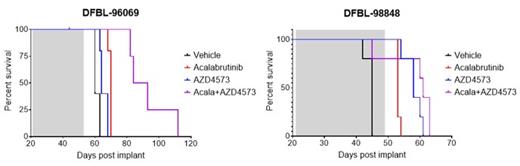
Most MCL cell lines are not responsive to BTK inhibition and, therefore, did not show combination benefit with AZD4573. We, therefore, chose to evaluate the in vivo activity of AZD4573 +/- acalabrutinib in 3 disseminated r/r MCL PDX models. In DFBL-44685, an acalabrutinib-unresponsive model harboring a CARD11 mutation, AZD4573 showed moderate activity, reducing MCL cells in all compartments analyzed (peripheral blood, bone marrow, and spleen) by >40% two weeks into treatment and increasing overall survival benefit (P<0.01). In an acalabrutinib-responsive MCL PDX, DFBL-96069, the combination of AZD4573 with acalabrutinib reduced tumor burden in all compartments analyzed by >80% and significantly increased survival over vehicle as well as monotherapy treatments (P<0.001). The third model, DFBL-98848, expresses high Bfl1. In this model, AZD4573 monotherapy reduced disease burden in all compartments analyzed by >50% and significantly increased survival over vehicle (P<0.001). Additionally, AZD4573 plus acalabrutinib further reduced disease burden and increased survival in this model compared to AZD4573 alone, although this did not reach statistical significance.
Our findings show that targeting CDK9 with AZD4573 can effectively induce apoptosis in a range of MCL cell lines and PDX models, including acalabrutinib-sensitive and -insensitive models as well as those expressing high levels of Bfl1. In 3 r/r MCL PDX models, single agent AZD4573 significantly reduced the tumor burden in the peripheral blood, bone marrow, and spleen of the affected mice, resulting in increased survival. Combination of AZD4573 with acalabrutinib resulted in greater anti-tumor activity than either monotherapy. Altogether, these data suggest that AZD4573, alone or in combination with acalabrutinib, could be an effective therapy for patients with r/r MCL.
Boiko: AstraZeneca: Current Employment, Current equity holder in publicly-traded company. Andersen: AstraZeneca: Current Employment, Current equity holder in publicly-traded company. Liang: AstraZeneca: Current Employment, Current equity holder in publicly-traded company. Dowdell: AstraZeneca: Current Employment, Current equity holder in publicly-traded company. Reimer: AstraZeneca: Current Employment, Current equity holder in publicly-traded company. Drew: AstraZeneca: Current Employment, Current equity holder in publicly-traded company. Townsley: AstraZeneca: Current Employment, Current equity holder in publicly-traded company. Cidado: AstraZeneca: Current Employment, Current equity holder in publicly-traded company.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal